

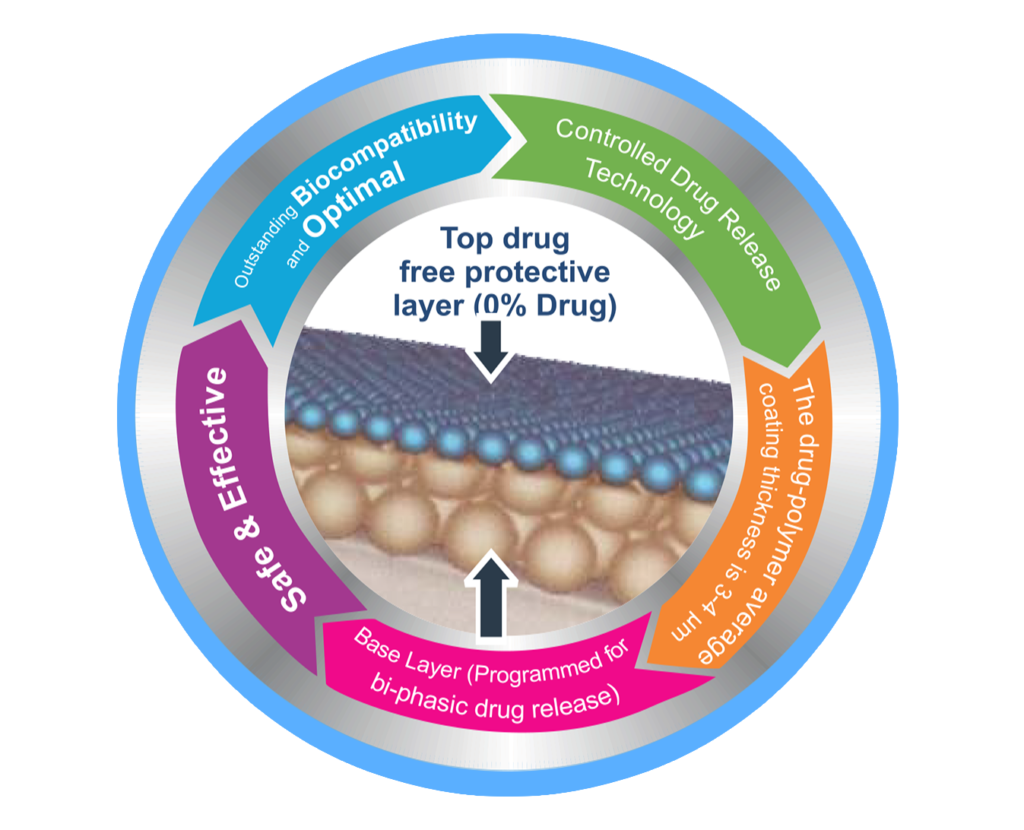


Unique Technology
Co-Cr stent platform has propriety unique electro polishing technology, which enables unsurpassed surface finishing to ensure an excellent deliverability, Even scaffolding & outstanding radial strength in NOVA MATRIX
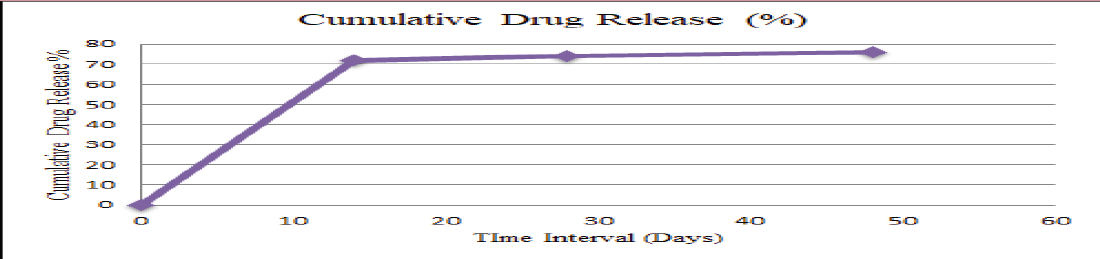
Proven drug release kinetics
NOVA MATRIX has proven drug release kinetics
Initial Burst Release of 40% – 50% sirolimus in the first week followed by susteined release upto 45-48 days to maintain therapeutic sirolimus drug concentration to optimise the antitissue proliferative action